Book Appointment Now

Stem Cell Therapy in Colombia: A Complete Medical Tourism Guide
Stem cell therapy in Colombia costs 50-70% less than US prices, with INVIMA-regulated GMP labs and clinics in Medellín and Bogotá serving 15,000+ American patients annually.
Treatments range from $3,000 for single joint injections to $25,000 for complex neurological protocols, compared to $10,000-$50,000+ in the US. Colombia’s WHO ranking of 22nd globally (highest in Latin America) supports a mature medical tourism infrastructure across Medellín, Bogotá, and Cali.
Medical Tourism Packages connects patients with INVIMA-registered clinics including BioXcellerator and Stem Cells Colombia in Medellín, verified for GMP laboratory certification, transparent cell count specifications, and structured post-treatment follow-up. Below, you will find evidence reviews by condition, protocol pricing, clinic comparisons, travel logistics, safety guidance, and answers to 14 frequently asked questions about stem cell therapy in Colombia.
What Is Stem Cell Therapy and How Does It Work?
Stem cell therapy uses specialized cells to support tissue repair and reduce inflammation. Most Colombian clinics use mesenchymal stem cells (MSCs) from umbilical cord tissue. These cells can reduce inflammation and modulate immune responses. The gap between laboratory results and real-world outcomes remains significant.
What Types of Stem Cells Do Colombian Clinics Use?
Colombian clinics primarily use allogeneic MSCs derived from umbilical cord Wharton’s Jelly. These donor cells require no harvesting surgery from the patient. Most facilities in Medellín and Bogotá prefer this source for convenience and consistency of supply.
Some clinics offer autologous therapy using a patient’s own bone marrow or adipose (fat) tissue. Autologous cells carry lower immune rejection risk. However, they require a minor harvesting procedure. Allogeneic cells from Wharton’s Jelly contain higher cell concentrations and may carry stronger anti-inflammatory properties.
Clinics use multiple delivery methods depending on your condition:
- Intra-articular injection: Cells injected directly into a joint. Used for knee, hip, and shoulder conditions. Lowest risk delivery method.
- IV infusion: Cells delivered through a drip. Used for systemic, autoimmune, and anti-aging protocols.
- Intrathecal injection: Cells injected into spinal fluid. Used for neurological conditions. Highest risk delivery method.
- Direct tissue injection: Cells injected into localized damaged tissue.
- Combination protocols: IV infusion paired with hyperbaric chamber sessions.
How Do Stem Cells Work to Repair Tissue?
MSCs work by releasing growth factors and signaling proteins that reduce inflammation and stimulate nearby cells to repair damaged tissue. They do not directly replace damaged cells in most cases. Their main effect is paracrine signaling, meaning they send chemical messages to surrounding cells.
MSCs can theoretically differentiate into bone, cartilage, and fat cells. In practice, most therapeutic benefit comes from their anti-inflammatory and immune-modulating effects. Clinics often describe “proprietary protocols” and “enhanced potency” processes. These claims rarely appear in peer-reviewed research. They are marketing terms for processes you cannot independently verify.
What Are the Known Risks and Side Effects of Stem Cell Therapy?
Stem cell therapy carries real risks, ranging from mild and temporary to severe and permanent. Risk level depends heavily on the delivery method used. Intrathecal (spinal) injections carry the highest risk. Intra-articular joint injections carry the lowest.
Common immediate side effects include:
- Joint pain and swelling at injection sites (most common, resolves in 48–72 hours)
- Flu-like symptoms including fatigue and low-grade fever
- Infection at injection or infusion sites
- Allergic reactions to donor cells
- Blood clots with IV delivery
Long-term risks are less understood and include potential tumor formation and severe immune reactions. Do not pursue stem cell therapy if you have active cancer, are pregnant or breastfeeding, have active infections, severe immune disorders, or serious bleeding disorders.
Why Do Patients Choose Colombia for Stem Cell Therapy?
Patients choose Colombia for stem cell therapy primarily because of significant cost savings, geographic accessibility from the US, and a developed medical infrastructure. Colombia ranks 22nd globally in WHO Healthcare Rankings, the highest position in Latin America. Over 15,000 Americans chose Colombia for medical care in 2025.
How Do Stem Cell Therapy Costs Compare Between Colombia, the US, and Canada?
Stem cell therapy in Colombia costs 50–70% less than comparable treatments in the United States. Entry-level joint protocols start around $2,850–$3,000. Complex neurological programs can reach $25,000. US prices for the same treatments range from $10,000 to over $50,000.
| Protocol Type | Colombia Cost (USD) | US Cost (USD) | Canada Cost (USD) | Savings vs. US |
|---|---|---|---|---|
| Single Joint (knee/hip) | $3,000–$12,000 | $10,000–$30,000 | $10,000–$25,000 | 50–70% |
| Systemic IV Protocol | $5,000–$18,000 | $15,000–$50,000 | $15,000–$40,000 | 50–70% |
| Neurological Protocol | $10,000–$25,000 | $25,000–$50,000+ | $20,000–$50,000 | 40–60% |
| Anti-Aging / Aesthetic | $3,500–$15,000 | $10,000–$30,000 | $8,000–$20,000 | 40–60% |
| Autoimmune / Complex | $5,000–$20,000 | $20,000–$50,000 | $15,000–$45,000 | 50–65% |
For additional context on Colombia’s medical tourism landscape compared to other destinations:
| Factor | Colombia | Mexico | Costa Rica | United States |
|---|---|---|---|---|
| Cost Range | $3,000–$25,000 | $3,000–$20,000 | $6,000–$30,000 | $10,000–$50,000+ |
| Regulation | Developing framework | Similar gaps | Limited oversight | FDA trials only |
| JCI Hospitals | 5–6 facilities | Extensive presence | Limited facilities | Established standards |
| English Support | Good in major cities | Variable | Strong | None needed |
| Flight Time (from Miami) | 3.5–4 hours | 2–4 hours | 3 hours | Domestic |
What Makes Colombia’s Healthcare Infrastructure Stand Out?
Colombia’s healthcare system ranks 22nd globally according to the World Health Organization, the top ranking in Latin America. The country is home to 40% of the top-ranked clinics and hospitals in Latin America. Medellín alone received 23,323 international patients in 2024, with 14% annual growth since 2010.
Colombia has 5–6 JCI-accredited hospitals. Fundación Santa Fe de Bogotá was the first Colombian hospital to earn JCI certification, in 2009, and maintains a collaboration agreement with Johns Hopkins Medicine International. Hospital Internacional de Colombia in Bucaramanga has earned the JCI Gold Seal six times and partners with the Mayo Clinic Care Network. Pablo Tobón Uribe Hospital in Medellín also holds JCI accreditation. Our comprehensive guide for American medical tourists in Colombia covers hospital options, travel logistics, and practical planning advice.
Important: JCI accreditation applies to overall hospital operations. It does not validate specific stem cell programs or regenerative medicine protocols. Patients must evaluate stem cell clinics independently from hospital accreditation status.
How Does Colombia’s Regulatory Environment Differ from the US?
Colombia’s regulatory environment for stem cell therapy differs from the US in one significant way: INVIMA, Colombia’s equivalent of the FDA, permits GMP-certified laboratories to culture and expand mesenchymal stem cells. The US FDA restricts this practice. This regulatory difference is a primary reason why Colombia has developed a stem cell therapy ecosystem.
Current INVIMA regulations (Decree 2493/2004, Resolution 5108/2005) cover tissue banking but not therapeutic applications of processed cells. INVIMA has acknowledged this gap. Commercial stem cell treatments operate as medical “procedures” regulated only at the tissue banking stage. A 2024 proposed law seeks a comprehensive framework for “terapias avanzadas” (advanced therapies), confirming that current oversight remains incomplete. Clinical trials supervised by INVIMA following international Good Clinical Practice standards represent the clearest regulatory pathway.
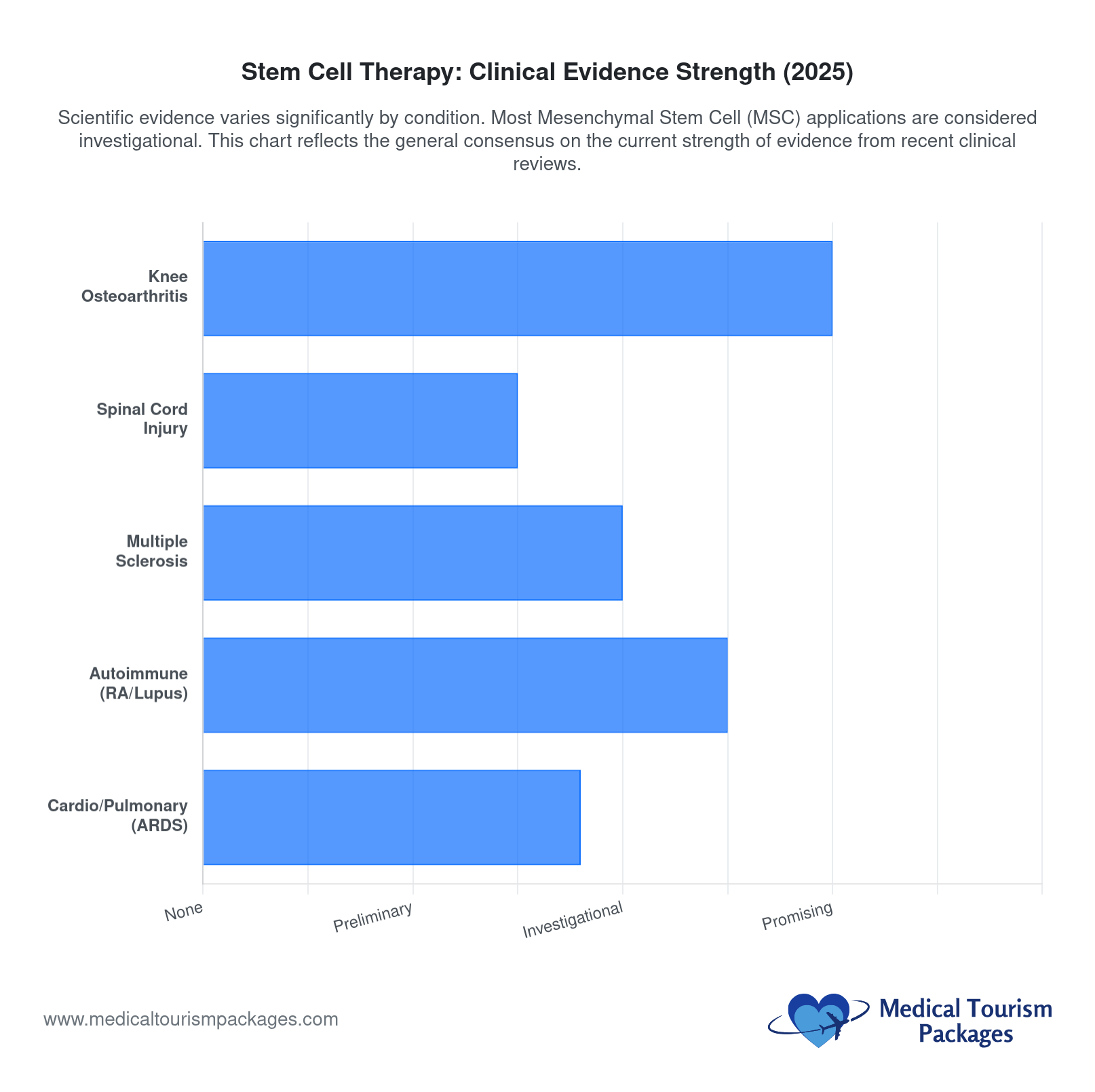
Which Conditions Have the Strongest Evidence for Stem Cell Therapy?
Evidence for stem cell therapy varies dramatically by condition. Some orthopedic applications have moderate support from randomized controlled trials. Most other applications, including neurological, autoimmune, and aesthetic conditions, remain in early investigational stages. The ISSCR (International Society for Stem Cell Research) considers most commercial offerings investigational at best.
What Does the Research Show for Orthopedic Conditions?
Knee osteoarthritis has the strongest evidence base for MSC therapy. A 2024–2025 meta-analysis of 8 randomized controlled trials with 502 patients found net positive effects on pain and symptoms. Improvements appeared in 12 of 15 RCTs relative to baseline. Adipose-derived MSCs at high doses showed the best results at six months.
Results are not guaranteed. Some high-quality trials found little to no pain relief at 3–6 months compared to placebo. Grade 3 knee osteoarthritis responds better than grade 4. MSC therapy appears most effective at slowing progression in earlier-stage disease. Benefits, when they occur, typically begin 4–12 weeks after treatment and peak at 3–6 months.
Sports injuries, back pain, and degenerative disc disease have limited evidence. Most data comes from case studies and small trials. Randomized controlled data is minimal for these applications.
What Is the Evidence for Neurological Conditions?
Neurological applications carry very low evidence and very high risk. Intrathecal injection, which delivers cells directly into spinal fluid, is the delivery method used for most neurological protocols. It is the highest-risk route available. Potential complications include infection, bleeding, headache, and spinal cord injury.
For progressive multiple sclerosis, small cohort trials have shown decreased MRI brain lesions in some patients. These are pilot studies with very small patient numbers. No established efficacy exists for MS, Parkinson’s disease, traumatic brain injury, spinal cord injury, or cerebral palsy. All neurological stem cell applications are investigational only. Exercise extreme caution before pursuing any spinal delivery protocol.
What Does the Research Show for Autoimmune and Other Conditions?
Autoimmune conditions show early signals but no confirmed efficacy. A 2025 global review published in Frontiers in Immunology analyzed 244 MSC trials for autoimmune diseases. It found that 83.6% of trials were in early Phase I–II stages. The most studied conditions were Crohn’s disease (85 trials), systemic lupus erythematosus (36 trials), and scleroderma (32 trials).
For cardiovascular conditions, a 2024–2025 comprehensive review of 27 clinical trials found acceptable safety profiles but unconfirmed efficacy. MSC-based therapy is the most widely used approach in cardiovascular research. Long-term oncogenic risk remains a concern, though no cancer cases have been directly linked to MSC treatment in recent trials.
Aesthetic and anti-aging applications (hair restoration, facial rejuvenation, wrinkle reduction) rely mostly on testimonials and before/after photos. No rigorous trial data supports these applications. Sexual health applications (erectile dysfunction, vaginal rejuvenation) have very limited data from small studies with unclear long-term effects. Patients comparing treatment options across the region can also explore stem cell therapy options throughout Latin America to understand how Colombia’s offerings compare to other destinations.
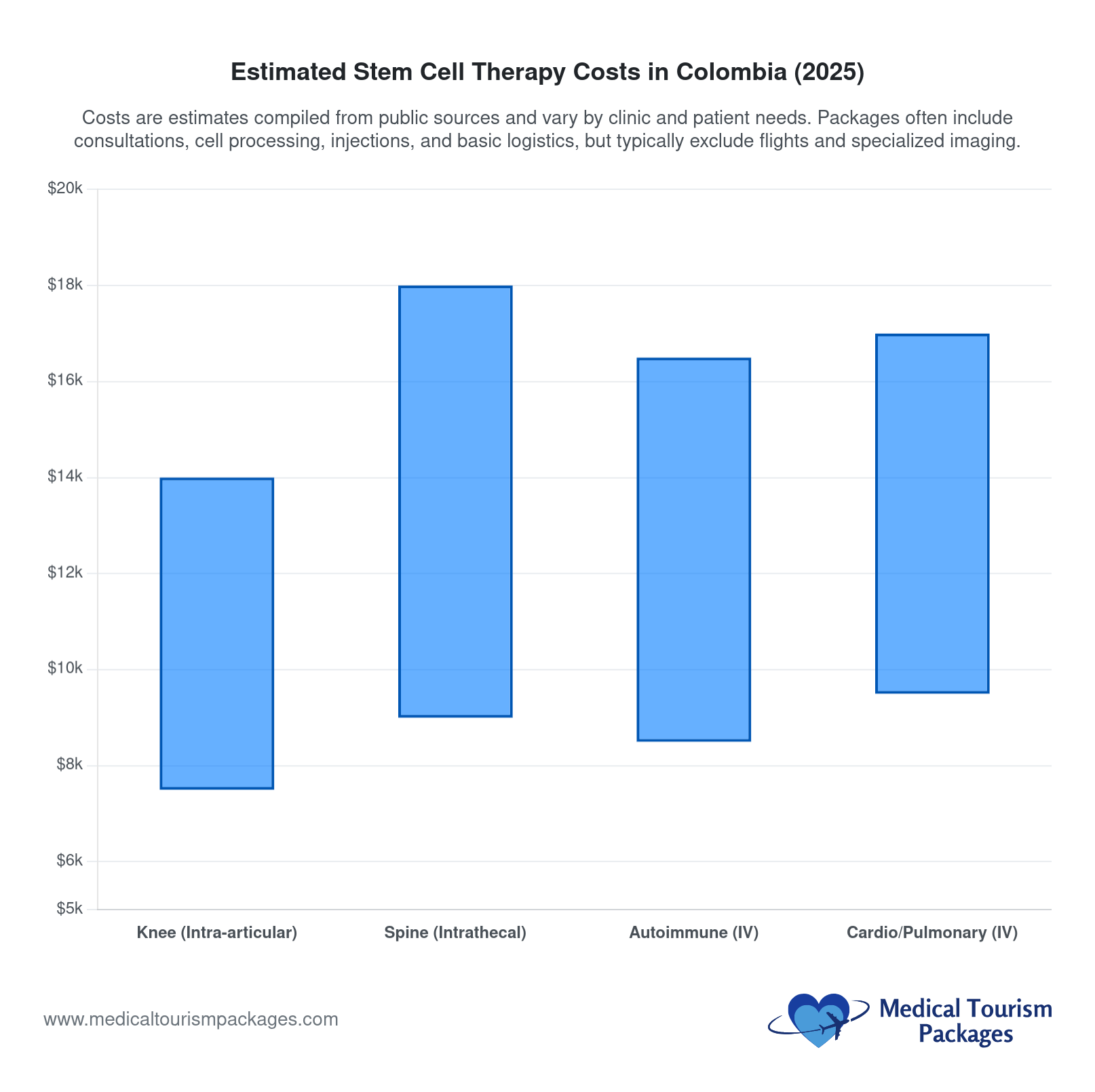
How Much Does Stem Cell Therapy Cost in Colombia?
Stem cell therapy in Colombia costs $2,850–$25,000+ depending on protocol complexity, cell counts, and included services. Entry-level joint injection packages start around $2,850 (Bookimed, February 2026). Multi-session neurological protocols reach $25,000 or more. The exchange rate is approximately 3,764 Colombian pesos per US dollar (March 2026).
What Do Different Stem Cell Protocols Cost in Colombia?
Protocol pricing in Colombia varies by treatment type, cell counts, and number of sessions:
| Treatment Type | Colombia Price Range (USD) | Typical Cell Count | Typical Stay |
|---|---|---|---|
| Single Joint (knee, hip, shoulder) | $3,000–$12,000 | 20–100 million MSCs | 3–5 nights |
| Systemic IV Protocol | $5,000–$18,000 | 100–300 million MSCs | 5–7 nights |
| Neurological Protocol | $10,000–$25,000+ | Multiple infusions | 10–14 nights |
| Anti-Aging / Aesthetic | $3,500–$15,000 | 50–200 million MSCs | 3–7 nights |
| Autoimmune / Complex | $5,000–$20,000 | 150–350 million MSCs | 7–14 nights |
What Does a Typical Stem Cell Package Include?
Most packages include cell processing, the treatment procedure, pre-treatment consultations, hotel accommodation, VIP airport transfer, and monitoring during your stay. Most packages exclude international flights and several significant costs.
Typically included in packages:
- Cell processing, preparation, and quality testing
- Injection or infusion procedure
- Pre-treatment consultations
- Hotel accommodation (3–14 nights depending on protocol)
- VIP airport transfer
- Post-treatment monitoring during your stay
Budget separately for these costs not included in most packages:
- International flights: $500–$2,000+ round trip
- Pre-treatment imaging: MRI or CT scans
- Laboratory tests: Comprehensive pre-treatment bloodwork
- Travel insurance with medical evacuation: $200–$800 (minimum $100,000 evacuation coverage recommended)
- Companion expenses: Accommodation and meals for travel partners
- Post-treatment rehabilitation: Physical therapy after returning home
- Complication management: Any additional care required for adverse events
Payment terms typically require a 10–30% deposit at booking. The balance is due on arrival. Credit cards are widely accepted and provide dispute protection. Many facilities prefer wire transfers for large payments. US dollars are accepted at some clinics, but converting to Colombian pesos yields better rates.
How Do Prices Vary Between Clinics and Why?
Prices vary because of differences in cell processing complexity, facility overhead, cell counts used per protocol, marketing spend, and what is bundled in the package. A clinic charging $12,000 for a joint injection is not necessarily offering a better product than one charging $5,000. The difference may reflect a nicer hotel, more included services, or higher marketing costs.
Price is not a reliable quality indicator in this market. Focus instead on verifiable quality factors: GMP lab certification, cell viability percentages, exact cell counts, and INVIMA registration status. Some clinics charge premiums for brand recognition rather than superior clinical protocols.
How Do You Choose a Safe Stem Cell Clinic in Colombia?
You choose a safe stem cell clinic in Colombia by verifying regulatory documentation, laboratory quality certifications, published outcome data, and transparent informed consent processes. GMP laboratory certification is the minimum standard for cell processing. Cell viability above 85% and exact cell counts (20–300 million depending on application) are quality benchmarks to request in writing.
What Documentation Should You Verify Before Booking Any Clinic?
Before booking, request and verify these items from any clinic:
Regulatory compliance (non-negotiable):
- INVIMA registration number and current status (verify independently at invima.gov.co)
- Clinical trial registration number (NCT number) if applicable
- Independent ethics committee approval documentation
- GMP certification for the cell processing laboratory
- Professional liability insurance verification
Clinical evidence and safety:
- Published studies on their specific cell product
- Documented success and complication rates for your condition
- Clear risk profiles with detailed informed consent documents
- Patient follow-up protocols and documentation
- Systematic adverse event reporting processes
Treatment quality:
- Exact cell count specifications (20–300 million depending on application)
- Cell viability percentage (must exceed 85%)
- Donor screening and safety testing documentation
- Sterility and quality control testing documentation
- Emergency protocols for adverse reactions
Business practices:
- Fully itemized quotes with all potential costs listed
- Clear policies for treatment failures or complications
- Coordination protocols with your home physicians
- Medical records privacy and patient data protection policies
- Post-treatment emergency support and accessibility
What Red Flags Indicate an Unsafe Clinic?
Red flags indicate clinics that prioritize revenue over patient safety. Avoid any clinic that shows these warning signs:
- Cure claims: Promises of cures or guaranteed results for any condition
- Missing documentation: Cannot provide INVIMA registration or GMP certification
- Excessive condition lists: Claims to treat dozens of conditions without condition-specific evidence
- High-pressure sales: Pressure to decide quickly or pay deposits immediately
- Physician isolation: Refuses to communicate with your home doctor
- No follow-up system: No structured post-treatment monitoring or emergency protocols
- Vague cell specs: Cannot provide exact cell counts or viability percentages in writing
Which Stem Cell Clinics Operate in Medellín, Bogotá, and Cali?
Medellín is the primary hub for stem cell clinics in Colombia. Bogotá has the highest concentration of JCI-accredited general hospitals but fewer dedicated stem cell clinics. Cali has a smaller commercial stem cell market.
BioXcellerator in Medellín is the most internationally recognized stem cell clinic in Colombia. It operates an onsite GMP-certified laboratory with full control over cell testing, screening, purification, and expansion. BioXcellerator has a recognized working relationship with INVIMA and received approval for a clinical trial using MSCs for COVID-19 treatment. The clinic operates within an integrated complex that includes hotel accommodation and dining. It tracks patients for 12–19 months post-treatment and self-reports positive results in approximately 80–90% of patients. This figure is self-reported and has not been independently validated.
Stem Cells Colombia, also in Medellín, specializes in orthopedic and systemic IV protocols. The clinic uses 150–350 million cells per protocol, which is higher than many competitors. It offers concierge-style coordination including airport pickup and hotel arrangements. The clinic is INVIMA-registered and works with GMP and ISO-standard laboratories.
MatrixCell in Bogotá is a regenerative medicine specialist clinic. Stem Cells Kyron Colombia in Cali describes itself as a pioneer in regenerative medicine in that city. For patients specifically researching blood disorder treatments, see our overview of stem cell transplants in Colombia, which covers hematopoietic stem cell transplantation at accredited hematology centers.
| Facility | City | JCI | GMP Lab | International Affiliation |
|---|---|---|---|---|
| Fundación Santa Fe de Bogotá | Bogotá | Yes (since 2009) | N/A (general hospital) | Johns Hopkins Medicine International |
| Hospital Internacional de Colombia | Bucaramanga | Yes (6x) | N/A (general hospital) | Mayo Clinic Care Network |
| Fundación Valle del Lili | Cali | Yes | N/A (general hospital) | None noted |
| Pablo Tobón Uribe Hospital | Medellín | Yes | N/A (general hospital) | None noted |
| BioXcellerator | Medellín | No | Yes (INVIMA GMP) | US-based leadership team |
| Stem Cells Colombia | Medellín | No | Yes (GMP/ISO) | None noted |
What Do You Need to Know About Traveling to Colombia for Treatment?
Traveling to Colombia for stem cell therapy requires planning for visa requirements, flight logistics, accommodation in safe neighborhoods, and specific safety precautions. The US State Department currently rates Colombia at Level 3: Reconsider Travel, issued April 2025. This is the second-highest warning level. Plan accordingly.
What Are the Visa and Entry Requirements for US and Canadian Patients?
US and Canadian citizens do not need a visa for tourism in Colombia. Both nationalities enter visa-free for up to 90 days. Requirements differ by nationality:
US citizens:
- No visa required for stays up to 90 days
- Extendable to 180 days total per calendar year
- Must show proof of onward or return travel
- Must show proof of sufficient funds
- Valid passport required (check minimum validity requirements with your airline)
Canadian citizens:
- No visa required for stays up to 90 days
- Entry fee of 256,000 COP (approximately CAD $85) payable on arrival
- Must complete the “Check Mig” online form before arrival and before departure through airports
- Valid passport required
Yellow fever vaccination certificate is required for travelers visiting endemic regions of Colombia. Check which areas you plan to visit before travel.
How Do You Get to Medellín or Bogotá from the United States?
You can reach Medellín and Bogotá with direct flights from several major US cities. Miami and Houston have the shortest flight times. Los Angeles requires a connecting flight. Medellín’s José María Córdova Airport (MDE) and Bogotá’s El Dorado International Airport (BOG) are the primary entry points.
| Origin City | Destination | Airlines | Approx. Flight Time | Approx. Round-Trip Cost |
|---|---|---|---|---|
| Miami | Medellín (MDE) | Avianca, American, Copa | ~3.5 hours | $265–$500 |
| Miami | Bogotá (BOG) | Avianca, American, Copa | ~3.5–4 hours | $300–$600 |
| Houston | Medellín / Bogotá | United, Copa, Avianca | ~4–5 hours | $350–$700 |
| New York (JFK/EWR) | Medellín | Avianca, Copa | ~5–6 hours (direct) | $400–$800 |
| Los Angeles | Bogotá / Medellín | Copa, Avianca (via hub) | ~8–10 hours (connecting) | $377–$900 |
Which Neighborhoods Are Best for Medical Tourists in Bogotá and Medellín?
Stay in established northern neighborhoods in Bogotá and in El Poblado in Medellín. These areas have concentrated medical facilities, security infrastructure, and English-speaking services suited for international patients recovering from treatment.
In Bogotá, the safest neighborhoods for medical tourists are Usaquén, El Chicó, Rosales, and Zona Rosa (also called Zona T). These are in the northern part of the city. Bogotá has the highest concentration of JCI-accredited hospitals in Colombia. It is the best choice for complex cases requiring high-level hospital infrastructure. The city sits at 2,640 meters elevation, which may require a day or two for acclimatization.
In Medellín, El Poblado is the primary neighborhood for medical tourists. It has modern hotels, restaurants, and walking-distance amenities. The area is well-patrolled and popular with international patients. Laureles is a quieter residential option preferred by long-stay patients. Medellín sits at 1,500 meters elevation with a temperate “City of Eternal Spring” climate ideal for recovery.
In Cali, the medical tourism infrastructure is smaller with fewer direct international flights. Cali is suitable for patients specifically seeking treatment at Fundación Valle del Lili or Cali-based clinics.
What Safety Precautions Should You Take in Colombia?
Colombia carries a US State Department Level 3 “Reconsider Travel” advisory as of April 2025. This is the second-highest warning level. Medical tourists should take specific precautions beyond standard travel safety.
The US Embassy in Bogotá has issued a specific alert about a rise in crimes involving scopolamine (also called “Devil’s Breath”). This drug is used to render victims compliant and erase their memory. Criminals administer it through drinks, food, and even direct skin contact. Dating apps have also been used to lure foreigners before drugging them.
Essential safety precautions for medical tourists in Colombia:
- Never leave a drink unattended at any venue
- Never accept food, drinks, or substances from strangers or new acquaintances
- Use Uber or InDriver instead of street taxis
- Use ATMs inside banks or malls during daylight hours only
- Avoid displaying valuables, jewelry, or electronics in public
- Keep a low profile and avoid drawing attention to yourself as a foreign visitor
- Exercise extreme caution with online dating apps during your stay
Emergency contacts for Colombia:
- US Embassy Bogotá: (+57) (601) 275-2000 | Carrera 45 No. 24B-27
- US Embassy after-hours emergency: +(57) (1) 275-4021
- From US/Canada: +1-888-407-4747
- Colombia emergency services: 123 (police, fire, ambulance)
- Tourist police: Available in major cities. Look for “Policía de Turismo.”
What Are the Risks and How Should You Handle Emergencies?
Stem cell therapy risks range from mild soreness to potentially life-threatening complications depending on delivery method. Joint injections carry the lowest risk. Spinal (intrathecal) injections carry the highest risk. Long-term risks, including tumor formation, remain incompletely understood across all delivery methods.
What Are the Risks for Each Type of Stem Cell Delivery?
Risk level varies significantly by how cells are delivered:
| Delivery Method | Common Risks | Serious Risks | Risk Level |
|---|---|---|---|
| Intra-articular (joint injection) | Pain, swelling at injection site | Infection, cartilage damage (rare) | Lowest |
| IV infusion | Flu-like symptoms, fatigue, fever | Blood clots, severe allergic reaction | Moderate |
| Intrathecal (spinal) | Headache, back pain | Infection, bleeding, neurological injury, death | Highest |
| Direct tissue injection | Local pain, swelling | Infection, local tissue reaction | Low to moderate |
Long-term risks across all delivery methods include potential tumor formation, uncontrolled cell growth, autoimmune reactions, and unknown effects on fertility. These risks are not yet fully understood because long-term follow-up data remains limited.
What Does the Treatment Timeline and Recovery Process Look Like?
The treatment timeline spans weeks before travel through months after returning home. Most protocols require 3–14 days in Colombia depending on complexity. Simple joint injection protocols require 3–5 days. Neurological or multi-session protocols require 10–14 days or more.
Pre-travel preparation (4–8 weeks before):
- Virtual consultation with chosen clinic
- Submit medical records and imaging
- Get clearance from your home physician
- Complete required laboratory tests
- Purchase travel insurance with medical evacuation coverage
- Book flexible airfare
During your stay:
- Days 1–2: Airport pickup, hotel orientation, comprehensive evaluation, final imaging and labs, informed consent review
- Days 3–4: Cell preparation (24–48 hours required), treatment day with 2–4 hours of monitoring, immediate recovery with activity restrictions
- Remaining days: Daily check-ins, rest, and early observation for adverse reactions
After returning home:
- Weekly consultations with clinic during month 1
- Formal outcome assessments at 2–3 months and 6+ months
- Coordinate with home healthcare providers familiar with regenerative medicine
Set realistic expectations. Benefits, if they occur at all, typically begin 4–12 weeks post-treatment. Peak effects, if any, appear at 3–6 months. Not all patients respond. Some experience no change. Some experience temporary worsening.
How Should You Handle Insurance and Potential Emergencies?
US health insurance does not cover experimental treatments abroad or complications from them. You are responsible for all costs. Medical evacuation from Colombia can exceed $250,000. At minimum, purchase travel insurance with $100,000 in emergency medical evacuation coverage. Comprehensive travel insurance with evacuation coverage costs approximately $200–$800.
Emergency escalation protocol:
- Contact your clinic first for new symptoms at injection sites, recovery questions, or mild concerns
- Seek local emergency care immediately for high fever, neurological symptoms, chest pain, breathing difficulties, or signs of stroke
- Establish a 24/7 support contact with your clinic before treatment begins
- Carry your full treatment records at all times during and after your trip
After returning home, inform your primary care physician and any relevant specialists about the experimental therapy you received. They need to know what was done, what cells were used, and what to monitor for in the months following treatment.
What Key Terms Should You Know Before Choosing Treatment?
Understanding these terms will help you evaluate clinic claims and ask better questions during consultations.
What Are the Most Important Terms to Understand?
Mesenchymal Stem Cells (MSCs): Adult stem cells found in bone marrow, adipose tissue, and umbilical cord tissue. They can differentiate into bone, cartilage, and fat cells. Their primary therapeutic effect comes from anti-inflammatory signaling rather than direct replacement of damaged cells. Most Colombian clinics use MSCs as their cell source.
Allogeneic: Stem cells sourced from a donor rather than the patient. Most Colombian clinics use allogeneic cells from umbilical cord Wharton’s Jelly. These cells require no patient harvesting surgery but carry a slightly higher immune reaction risk than autologous cells.
Autologous: Stem cells sourced from the patient’s own body, typically from bone marrow or fat tissue. Lower immune rejection risk but requires a harvesting procedure. Some Colombian clinics offer this option.
Wharton’s Jelly / Umbilical Cord Tissue: The primary source of MSCs for most Colombian clinics. Wharton’s Jelly is a gelatinous substance in the umbilical cord. It contains high concentrations of MSCs that can be collected without harm to the donor or infant.
GMP (Good Manufacturing Practices): International quality standards for pharmaceutical and cell processing facilities. A GMP-certified laboratory meets defined standards for sterility, consistency, and documentation. GMP certification is the minimum quality standard to require from any clinic.
INVIMA: Colombia’s Instituto Nacional de Vigilancia de Medicamentos y Alimentos, the national regulatory authority equivalent to the US FDA. INVIMA regulates tissue banking but has acknowledged that therapeutic use of processed cells falls outside its current regulatory categories.
JCI (Joint Commission International): International healthcare accreditation organization. JCI certification indicates high-quality hospital operations and management standards. It does not validate specific stem cell protocols or regenerative medicine programs.
Intrathecal: Delivery of cells directly into cerebrospinal fluid via spinal tap. The highest-risk route for stem cell delivery. Used for neurological conditions. Carries risks of infection, bleeding, headache, and spinal cord injury.
ISSCR (International Society for Stem Cell Research): The leading global organization providing scientific and ethical guidelines for stem cell research and clinical applications. The ISSCR considers most commercial stem cell offerings investigational. Their guidelines are a useful benchmark for evaluating clinic claims.
Frequently Asked Questions
How effective are stem cell treatments for my condition?
Effectiveness varies by condition. Knee osteoarthritis has the strongest support, with multiple 2024–2025 meta-analyses showing positive effects in most randomized trials. Conditions like Alzheimer’s, ALS, and autism have minimal evidence. Most commercial stem cell therapies remain investigational, with benefits unproven in rigorous trials.
How long before I see results?
If benefits occur, they usually begin 4–12 weeks after treatment, often peaking around 3–6 months. Some patients note improvement up to 12 months after treatment. Others see no change or a temporary worsening. Do not expect immediate results.
What’s the success rate?
Most clinics do not publish comprehensive, standardized outcome data. Reported success varies widely by condition, patient factors, and how “success” is defined. Many patients experience partial improvement rather than dramatic change. BioXcellerator self-reports positive results in approximately 80–90% of patients, but this figure is not independently validated.
Are multiple treatments necessary?
It depends on your condition and response. Some patients do well with a single treatment. Others may need additional sessions. Be cautious of clinics that push costly series upfront without allowing time to assess initial results. Allow 3–6 months to evaluate your response before considering additional treatment.
Is stem cell therapy safe?
Stem cell therapy is generally low risk for joint injections but carries real risks for IV and spinal delivery. Immediate risks include infection and allergic reactions. Long-term risks, including tumor formation and immune dysfunction, are not fully understood. Risk varies significantly by delivery method. Spinal injections carry the highest risk.
What are the most common side effects?
Common side effects include flu-like symptoms (fatigue, low-grade fever), localized pain and swelling at injection sites, and temporary worsening of baseline symptoms. These usually resolve within 48–72 hours. Serious but less common complications include severe allergic reactions, infections, and systemic immune responses.
Can stem cell therapy make my condition worse?
Yes. Though uncommon, treatments can worsen an underlying condition, trigger autoimmune reactions, or mask disease progression. Temporary worsening of symptoms in the first 2–4 weeks after treatment is relatively common. Careful candidate selection and ongoing monitoring are essential to manage this risk.
Are treatments approved by Colombian health authorities?
Most commercial stem cell treatments operate in a regulatory grey area. INVIMA regulates tissue banking under Decree 2493/2004 but has acknowledged that therapeutic use of processed cells falls outside existing categories. A 2024 proposed law for “terapias avanzadas” confirms this gap. INVIMA does permit GMP labs to culture and expand MSCs, unlike the US FDA.
Will my insurance cover complications from treatment in Colombia?
Most US health plans do not cover experimental treatments abroad or their complications. Medical evacuation from Colombia can exceed $250,000. Purchase travel insurance with at least $100,000 in emergency medical evacuation coverage before you travel. Verify your specific policy exclusions with your insurer before booking.
How do I choose a reputable clinic?
Use a 20-point vetting checklist. Prioritize clinics with INVIMA registration, GMP lab certification, cell viability above 85%, published outcome data, clear informed consent documents, and a post-treatment monitoring protocol. Avoid facilities that make cure claims or pressure quick decisions.
What type of stem cells are used in Colombian clinics?
Most Colombian clinics use allogeneic mesenchymal stem cells (MSCs) derived from umbilical cord Wharton’s Jelly. These donor cells require no harvesting procedure from the patient. They provide high cell concentrations but carry a slightly higher immune reaction risk than autologous (patient’s own) cells derived from bone marrow or fat tissue.
Why are prices so variable between clinics?
Prices reflect cell processing complexity, cell counts used, facility overhead, bundled services, and marketing spend. Wide price ranges also reflect genuine quality variation. Price alone is not a reliable quality indicator. Focus on verifiable quality factors like GMP certification, cell viability percentages, and INVIMA registration.
How long should I stay in Colombia?
Plan 3–7 days for simpler joint injection protocols and 10–14 days for complex neurological or multi-session treatments. Build extra time into your schedule for possible complications or additional consultations. Arrive 1–2 days early to acclimatize, especially if traveling to Bogotá at 2,640 meters elevation.
What if I need emergency care after returning home?
Set up care with your home providers before you travel. Carry your full treatment records and clinic contact information at all times. Ensure your home medical team knows about the experimental therapy you received, which cells were used, and what complications to monitor for in the months following treatment.
Ready to Start Your Stem Cell Therapy Journey?
Medical Tourism Packages coordinates your entire healthcare journey to Colombia. We connect you with INVIMA-registered clinics and GMP-certified laboratories, arrange travel and accommodations in safe neighborhoods like El Poblado in Medellín or Usaquén in Bogotá, and provide bilingual support throughout your treatment and recovery.
Contact us today for a free consultation and personalized medical tourism package.